 They are in orbitals which are really just Orbits the way that planets are in orbit around the sun and we've talked about Right way to conceptualize how electrons or how they move or how they are distributed The outermost electron and we could call that the radius." That would work except for the fact that this is not the I just figure out the distance between the nucleus and That's easy to figure out the atomic radius. When they conceptualize an atom they imagine a positive nucleus with the protons in theĬenter right over here then they imagine theĮlectrons on these fixed orbits around that nucleus so they might imagine some electrons in this orbit right over here, just kind of orbiting around and then there might be a few more on this orbit out here orbiting around, orbiting around out here. Well what's the distance between the center of that circular object and the edge of it. If I'm trying to calculate the radius of some type of circular object I'm just thinking about Think well this might be a fairly straight-forward thing. 
Voiceover: Let's think a little bit about the notion of atomic size or atomic radius in this video. So each added proton adds a little more positive charge felt by the valence electrons than is shielded by the additional electrons. While they do provide some repulsion, they do not block out the entire positive of a proton. The newly added electrons going left to right in a period are being added to the same electron shell as the existing valence electrons. The net charge which electrons feel towards the nucleus after subtracting out the repulsions of the electrons is known as effective nuclear charge. The electrons orbit the nucleus in a variety of orbital shapes and do not completely shield other electrons from the nucleus. Which should make sense since atoms exist. The attractive force is stronger than the repulsive and so even the outermost electrons feel a net attraction to the nucleus. But these two conflicting forces, the attractive and repulsive forces, are not equal in magnitude. However, they also feel a repulsive force from the other electrons since they all possess the same negative charge. All electrons of an atom feel an attractive force to the nucleus because of the positive charge of the protons. To make the atoms neutral, an electron is also added to the atom. Is this trend also seen in other groups? On the diagram of the periodic table at the end of this worksheet, use arrows to show the trend in atomic radila a period and down a group.As we move right to left along the same period in the periodic table, the atomic number increases which means the number of protons in the nucleus also increases. How does atomic radius change down group 1 (Li to Fr)? 8. Is this trend also seen in other periods? 7. What is the general trend across the 3rd period (Na to Ne)? 6. To which group in the periodic table, do these elements belong? 5. Which elements have the smallest radii, represented by the lowest points of the graph? 4. To which group in the periodic table, do these elements belong? 3. 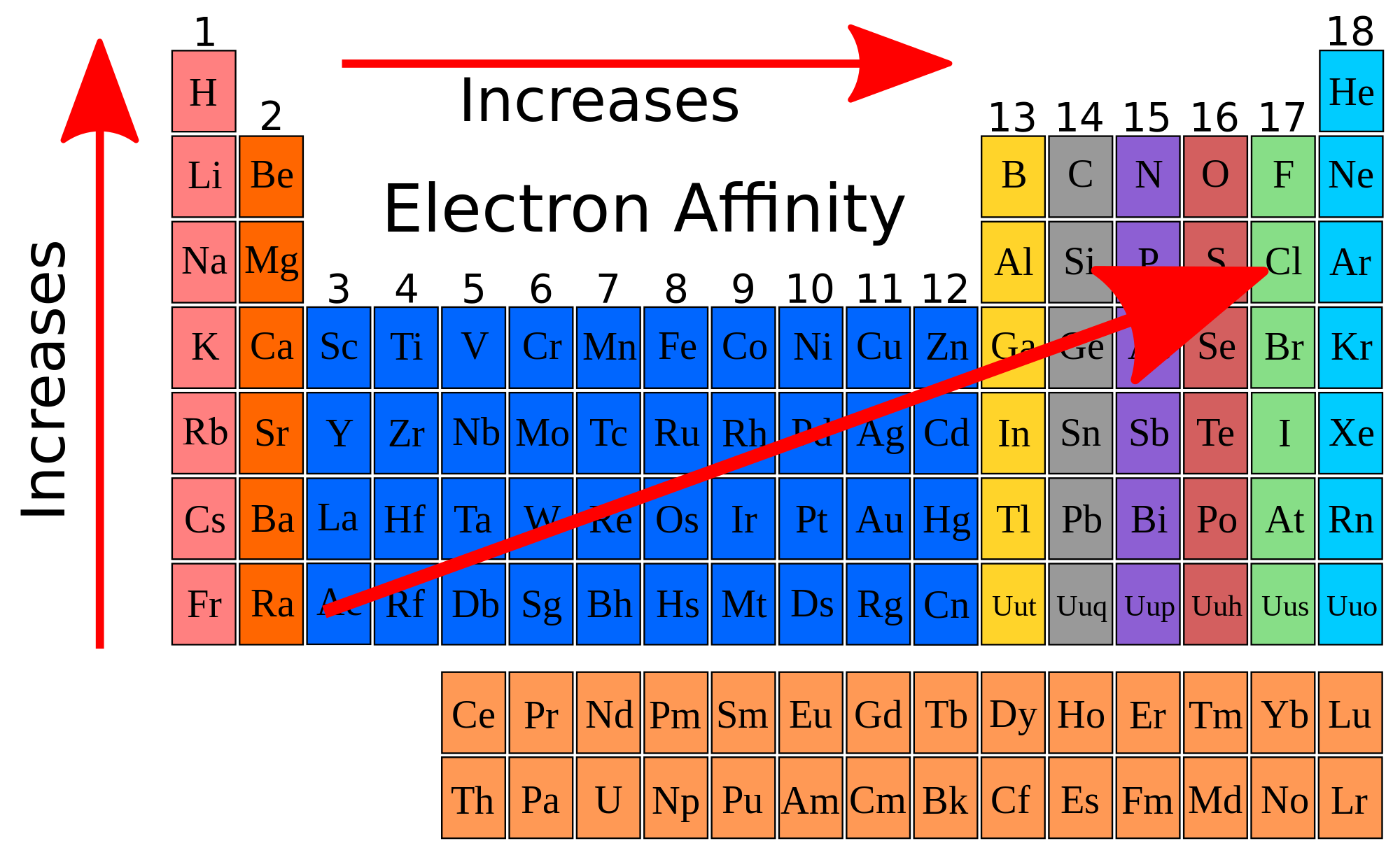
Which elements have the largest radii, represented by the tallest peaks? 2. Study the graph and answer the following questions lectron-electron repulsion Alone 1. Theses radii are often called covalent radil, or metallic radil (for metals) Here is a graph of atomic radius plotted against atomic number. This distance is called the bonding atomic radius and is shorter than the non-bonded radius. The distance between the nuclei of the bonding atoms is equal to two times the atomic radius. One way of measuring the atomic radius is to observe the distance between bonding atoms. This distance is very small so it is often reported in picometers (10 m) or angstroms (10-10 m). 
Atomic radius The radius of an atom describes the distance between the nucleus and the outermost electron. Some anomalies in these trends The reason behind the trend, effective nuclear charge.Periodic trends in atomic size (radius ionization energy and electron affinity.Periodic Trends in Physical Atomic Properties (Section 6.5 in the OpenStax text) After completing this worksheet you should know. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
